AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
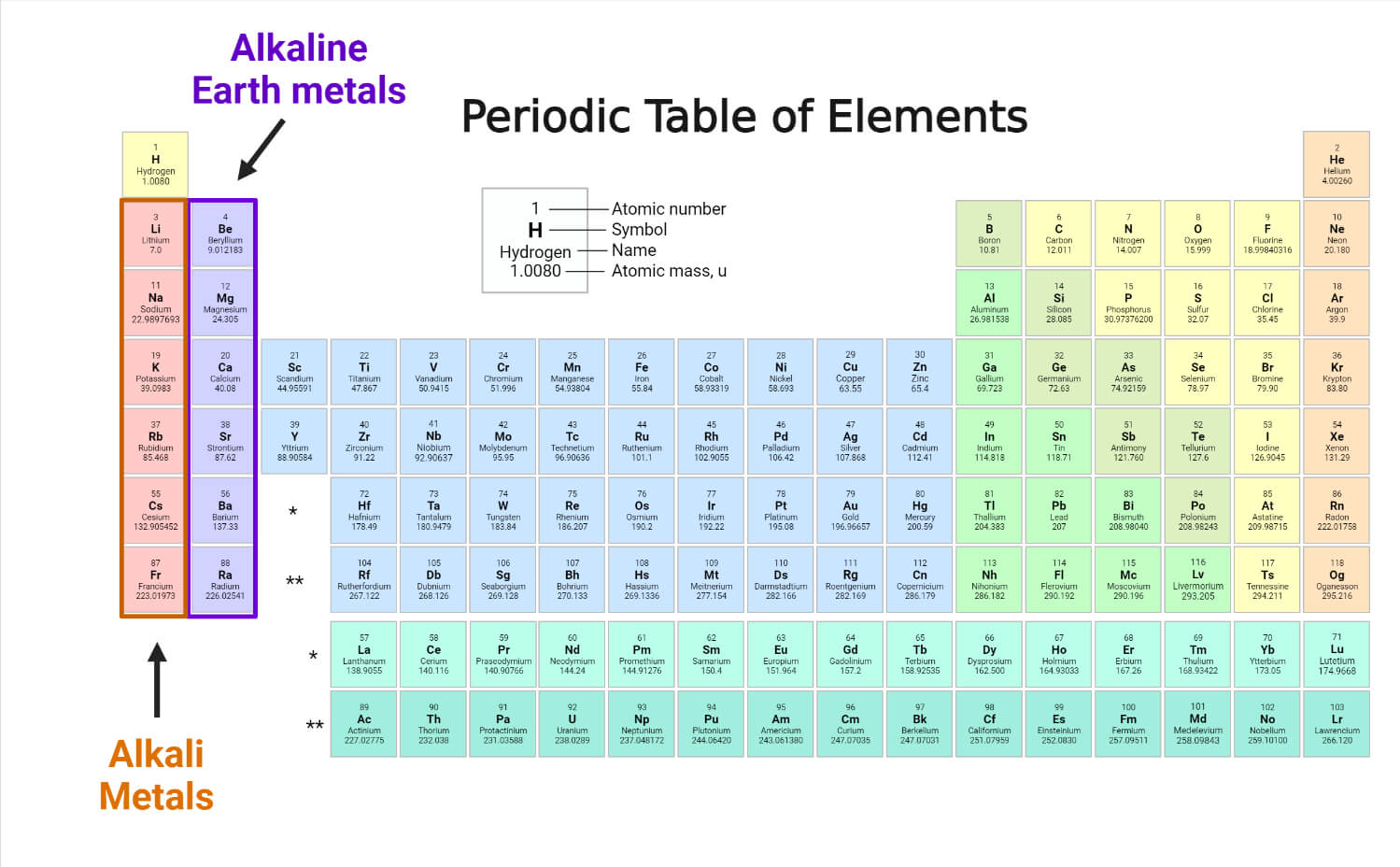
The nuclear radii of the Alkali Metals rise, going down the group. By and large, their densities rise while moving down the table, with the exemption that potassium is less thick than sodium. For example, while moving down the table, all the discovered and recognized Alkali Metals show expanding nuclear radius, diminishing Electronegativity, expanding reactivity, and diminishing, dissolving, and bubbling points just as heats of fusion and vaporization. The Alkali Metals are more like each other than the components in any of the other groups are to each other. Odd– odd primordial nuclides are uncommon in light of the fact that most odd– odd cores are exceedingly unstable with beta decay, on the grounds that the decay items are even-even, and are hence all the more emphatically bound, because of atomic blending impacts. Odd– odd cores have even mass numbers, while odd-even cores have odd mass numbers. Hence, in spite of the expectation, cesium is the most receptive of the Alkali Metals, not Francium.Īll the Alkali Metals have odd nuclear numbers thus, their isotopes must be either odd– odd (both proton and neutron numbers are odd) or odd-even (proton number is odd, however, neutron number is even). 
As opposed to the pattern of diminishing Electronegativities and Ionization energies of the Alkali Metals, francium's Electronegativity and Ionization energy are anticipated to be higher than cesium because of the relativistic adjustment of the 7s Electrons likewise, its nuclear span is relied upon to be strangely low. 
This impact likewise implies that most straightforward Lithium Alkalis are usually experienced in hydrated structure, on the grounds that the anhydrous structures are amazingly hygroscopic: this permits Alkalis like Lithium chloride and Lithium bromide to be utilized in dehumidifiers and forced air systems.įrancium is likewise anticipated to demonstrate a few contrasts because of its high nuclear weight, making its Electrons travel at significant parts of the speed of light and hence making relativistic impacts increasingly conspicuous. Conversely, Lithium perchlorate and other Lithium Alkalis with vast anions that can't be enraptured are considerably more steady than comparable to compounds of the other Alkali Metals, presumably in light of the fact that Li+ has high solvation energy. Lithium fluoride is the first Alkali Metal halide that is inadequately dissolvable in water, and Lithium hydroxide is the first Alkali Metal hydroxide that isn't deliquescent. Further, among their particular groups, just Lithium and magnesium structure organoMetallic compounds with critical covalent character (for example LiMe and MgMe2). For instance, Lithium frames a steady nitride, a property normal among all the soluble earth Metals (magnesium's group) however exceptional among the Alkali Metals. Lithium and magnesium have a diagonal relationship due to their comparable nuclear radii, with the goal that they demonstrate a few similitudes. The Chemistry of Lithium demonstrates a few contrasts from that of whatever is left of the group. What little that is thought about francium demonstrates that it is exceptionally close in conduct to cesium, not surprisingly? The physical properties of francium are much sketchier in light of the fact that the mass component has never been observed subsequently, any information that might be found in the writing is surely theoretical extrapolations. The Chemistry of francium isn't settled because of its very high level of radioactivity accordingly, the introduction of its properties here is constrained. Most of the Chemistry has been observed just for the first five individuals from the group. Their Chemistry is overwhelmed by the loss of their solitary valence Electron in the peripheral s-orbital to obtain the +1 oxidation state, because of the simplicity of ionizing this Electron and the high second ionization energy. The ns1 setup likewise results in the Alkali Metals having huge nuclear and ionic radii, just as high heat and electrical conductivity. They all solidify in the body-centered cubic precious stone structure and have particular fire hues on the grounds that their external Electron is all-around effectively excited. Henceforth, all the Alkali Metals are soft and have low densities, melting and bubbling points, and heats of sublimation, vaporization, and dissociation. The physical and chemical properties of the Alkali Metals can be promptly clarified by their having an ns1 valence Electron setup, which results in frail Metallic holding.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
